Health and Human Services Secretary Robert F. Kennedy Jr. announced the rescission of emergency use authorizations (EUAs) for COVID-19 vaccines, marking a significant shift in federal policy. RFK Jr. stated he fulfilled four key promises: ensuring vaccine availability for vulnerable populations, demanding placebo-controlled trials from pharmaceutical companies, and transitioning to full FDA approval for specific groups.
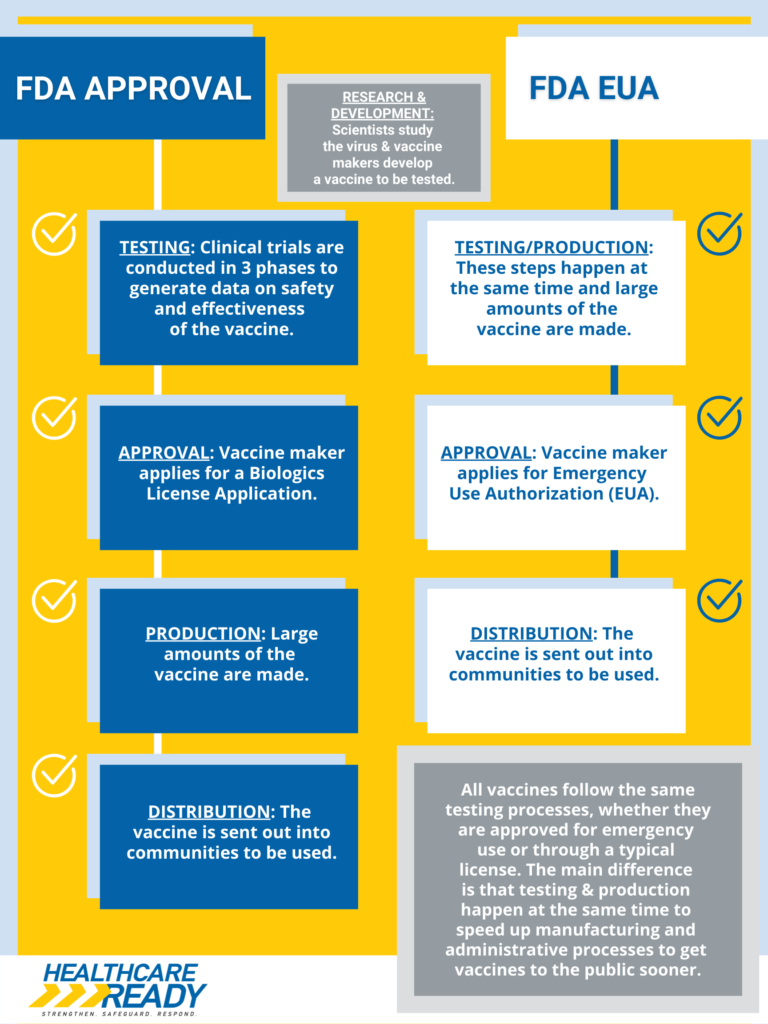
The decision follows the Biden administration’s termination of the national public health emergency on May 11, 2023. Under EUAs, medical products are authorized when no approved alternatives exist, but the standards differ from full FDA approval. The vaccines developed by Pfizer and Moderna initially received EUAs before securing full approvals in 2021 and 2022, respectively.
The FDA recently granted full approval for Moderna’s vaccine for children aged 6 months to 11 years, making it the first such option not reliant on an EUA. However, critics argue the move leaves gaps in access, particularly for healthy children. Dr. Susan Kressly of the American Academy of Pediatrics (AAP) called the decision “deeply troubling,” warning that barriers to vaccination could endanger children during respiratory virus season.
Kennedy’s actions include replacing the CDC’s vaccine advisory committee with members skeptical of COVID-19 vaccines, raising concerns about future coverage policies. While doctors may still prescribe vaccines off-label, this approach complicates access for many patients relying on pharmacies. The full implications of the policy shift remain under review.